The approved abbreviated new drug application (ANDA) is thereapuetically equivalent to the reference listed drug product Tegretol-XR ExtendedRelease Tablets, 100 mg, 200 mg, and 400 mg, of Novartis Pharmaceuticals Corporation, the company said.
The Carbamazepine extended-release tablets are indicated for use as an anti-convulsant drug and also for the treatment of the pain associated with true trigerminal neuralgia, Alembic Pharma said.
As per IQVIA, the tablets have an estimated market size of $71 million for the 1w months ending March 2025.
Alembic Pharma has a total of 225 ANDA approvals from the USFDA, including 202 final approvals and 23 tentative ones.
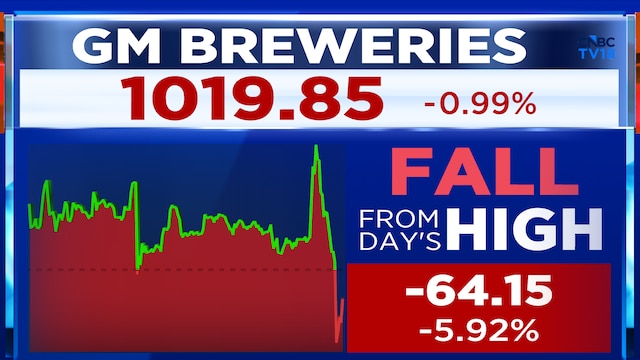
Alembic Pharma shares ended the previous session 1.91% lower at ₹999 apiece. The stock has declined 4.77% this year, so far.
Also Read: IndusInd Bank chief human resources officer Zubin Mody tenders his resignation